RECENT PAPER ON ICE CORE RECORDS MAY SUPPORT NATURAL TRANSPORT CYCLE FOR TRIFLUOROACETIC ACID
rifluoroacetic acid (TFA) is a naturally occurring substance and a ubiquitous component of the hydrosphere. It is found in fog snow and rain at levels in the 1990s that could not be explained by the known anthropogenic sources [1]. Annual TFA fluxes from pre-1990s ice core records may support the existence of a natural cycle transporting TFA from the oceans (which contain over 200 million tonnes of TFA) to the atmosphere where it can be rained out over land.
The recent paper [2] on ice core records of TFA and two short chain perfluorinated carboxylic acids (scPFCAs) reports annual deposition fluxes of TFA from 1967 for Mt. Oxford icefield (Ellesmere Island) and from 1977 for the Devon Ice Cap. Until the 1990s there were only limited anthropogenic sources for the introduction of TFA into the atmosphere and these do not appear to account for the deposition fluxes reported.
Prior to 1980, halothane anaesthetic was the dominant source of anthropogenic atmospheric TFA. The anaesthetic isoflurane was not introduced into clinical use until 1980 [3]. The molar yield of TFA from both anaesthetics is 0.6 [4]. The intermediate atmospheric breakdown product for halothane is CF3COCl, which is partially photolyzed reducing the yield of TFA [1]. By 1995 the annual production of halothane was estimated as a maximum of 1500 tonnes globally [1], resulting a TFA global yield of 520 tonnes [4]. The atmospheric lifetime of halothane (1.2 years) means that halothane will be well mixed in the atmosphere leading to local TFA deposition fluxes related to local precipitation.
The Devon Ice Cap is in a “polar desert” climatic region, where annual precipitation is often 200 mm, with minimal interannual variability [5]. Similar conditions occur for the Mt. Oxford icefield. This compares to global average precipitation of 980 mm/year [6]. From this, the annual TFA flux from halothane in the two ice core locations would have been about 0.2 µg/m2/yr [7].
Ice core records for TFA flux (µg/m2/yr for some years before 1980 [2]
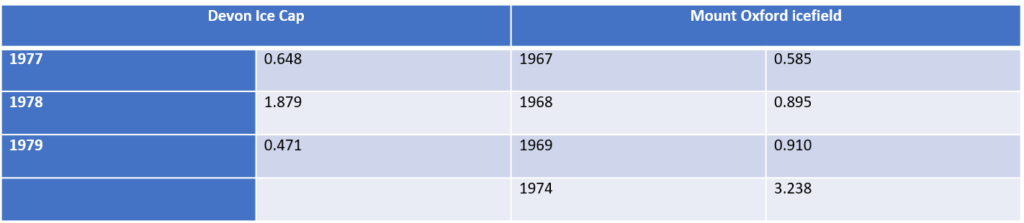
Ice core records for TFA flux (µg/m2/yr) are shown in the table for some years before 1980. The annual TFA fluxes from the ice core records are all higher or substantially higher than expected from anthropogenic sources in the period before 1980. The EFCTC paper “Transport of naturally occurring trifluoroacetic acid (TFA) by sea salt aerosol” [8] provides a possible explanation. The world’s oceans constitute a significant TFA reservoir (estimated at over 200 million tonnes) and the generation of sea salt aerosol by mechanical action of wind on the sea surface is well known. It is postulated that this provides a mechanism for transportation of significant quantities of TFA over considerable distances, further even than sea salt aerosol. This represents a variable background concentration of TFA in the environment that supplements any generated by decomposition of fluorocarbons. This background concentration has been shown to be highly variable and would depend on differences in generation of the sea salt aerosol, influenced by wind speed and temperature, differences in precipitation, with fog having a higher concentration than heavy rainfall, and differences in geographical location relative to the sea.
Consistent with natural TFA transportation by sea salt aerosol, a 2006 paper [9] concluded that open-water conditions in Baffin Bay during the stormy seasons (fall and spring) promote increased production, transport and deposition of sea-salt aerosols on Devon Ice Cap. The paper noted that increased wind fetch over open water results in more surface waves, which favours sea-spray production and concludes that most sea-salt aerosols deposited on Devon Ice Cap are likely to come from nearby, rather than distant, sources.
In conclusion, transport of natural TFA, from the global reservoir, by sea salt aerosol provides a possible explanation for the TFA annual flux at the Devon Ice Cap and Mt. Oxford icefield in the period before 1980 when anthropogenic sources in the atmosphere were very limited. Thus, there are background concentrations of TFA in the atmosphere, precipitation and surface water that augment any anthropogenic generation.
More information about TFA is available here.
References and notes
1. Boutonnet et al 1999 Environmental Risk Assessment of Trifluoroacetic Acid, Human and Ecological Risk Assessment · February 1999, DOI: 10.1080/10807039991289644 https://www.researchgate.net/publication/254217782
2. Ice core record of persistent short-chain fluorinated alkyl acids: Evidence of the impact from global environmental regulations; Heidi M. Pickard, Alison S. Criscitiello, Daniel Persaud, Christine Spencer, Derek C. G. Muir, Igor Lehnherr, Martin J. Sharp, Amila O. De Silva, Cora J. Young; Geophysical Research Letters First published: 23 April 2020 https://doi.org/10.1029/2020GL087535
3. History of anaesthesia society timeline http://www.histansoc.org.uk/timeline.html
4. IPCC/TEAP Special Report: Safeguarding the Ozone Layer and the Global Climate System chapter 2 page 154. Note that the ice core paper in its supplementary information reports that halothane has a TFA yield of 1, contrary to this reference and also Boutonnet 1999.
5. Influence of the Arctic Circumpolar Vortex on the Mass Balance of Canadian High Arctic Glaciers, A.S Gardner and M Sharp, Journal of Climate, 2006, 20, 4586
6. Global Precipitation: Means, Variations and Trends During the Satellite Era (1979–2014), R. F. Adler, G. Gu, M. Sapiano, Jian-Jian Wang, and G J. Huffman. Surv Geophys (2017) 38:679–699, DOI 10.1007/s10712-017-9416-4
7. The earth’s surface area is 510 million km2. Uniform TFA deposition of 520 tonnes would result in about 1 µg/m2/yr. Polar desert regions with about 20% of average global precipitation would have about 0.2 µg/m2/yr.
8. Available at https://www.fluorocarbons.org/wp-content/uploads/2020/04/EFCTC-Paper-Transport-of-natural-TFA-by-Sea-Salt-Aerosol-02.04.2020-1-1.pdf
9. Calibration of an ice-core glaciochemical (sea-salt) record with sea-ice variability in the Canadian Arctic, Christophe Kinnard, Christian M. Zdanpwicz, David A. Fisher, Cameron P. Wake, Annals of Glaciology 44(1):383-390 · November 2006 DOI: 10.3189/172756406781811349